Lung Ultrasound
Lung Ultrasound - How to Perform and Interpret
Lung ultrasound is a rapid, sensitive, and specific method for assessing the etiology of respiratory distress. For instance, in pulmonary edema, lung ultrasound (LUS) has been demonstrated to possess a higher diagnostic accuracy than the combination of physical examination and chest radiography. The learning curve for lung ultrasound is steep, allowing clinicians to acquire diagnostic proficiency relatively quickly.
Ultrasound imaging is based on the principle that when an ultrasound wave encounters a surface where acoustic impedance changes markedly, phenomena such as reflection, refraction, and scattering occur. The findings observed in lung ultrasound are explained by the interactions of ultrasound waves with air, interstitial fluid, and tissue. In contrast to most conventional ultrasound examinations, in which pathological findings can be directly visualized, many of the diagnostic features in lung ultrasound are infact artifacts created by these interactions between tissue and air. Nevertheless, some pathological changes in the lungs can also be directly visualized and interpreted without relying on artifacts.
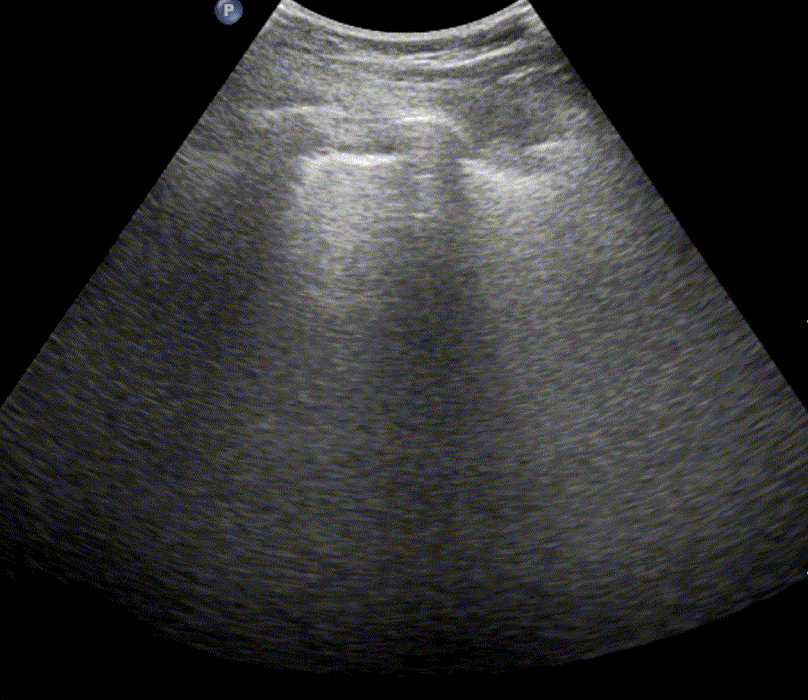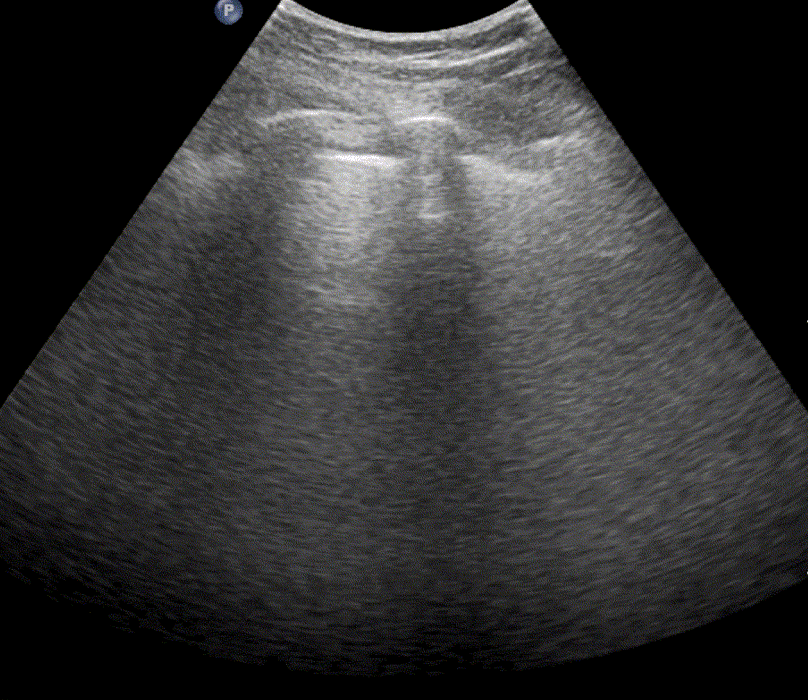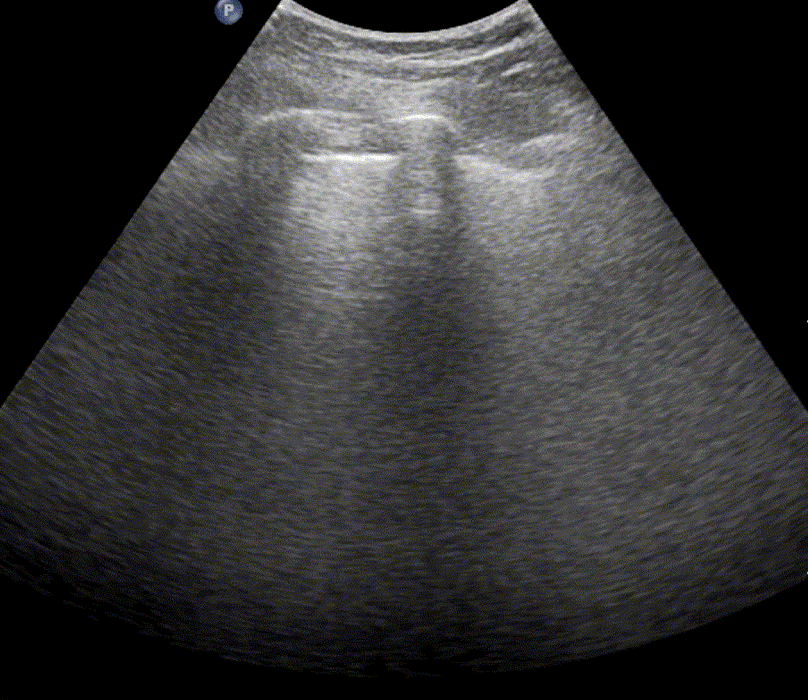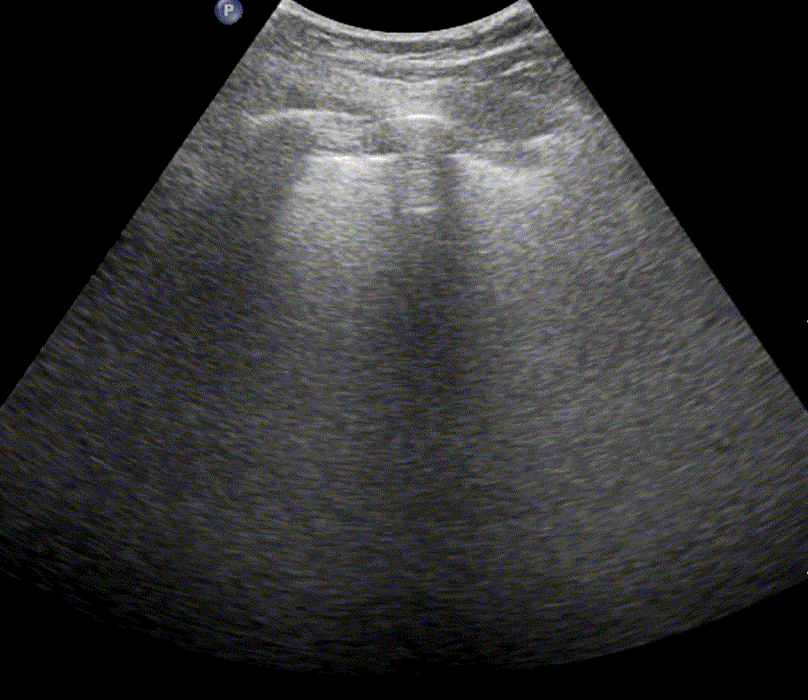
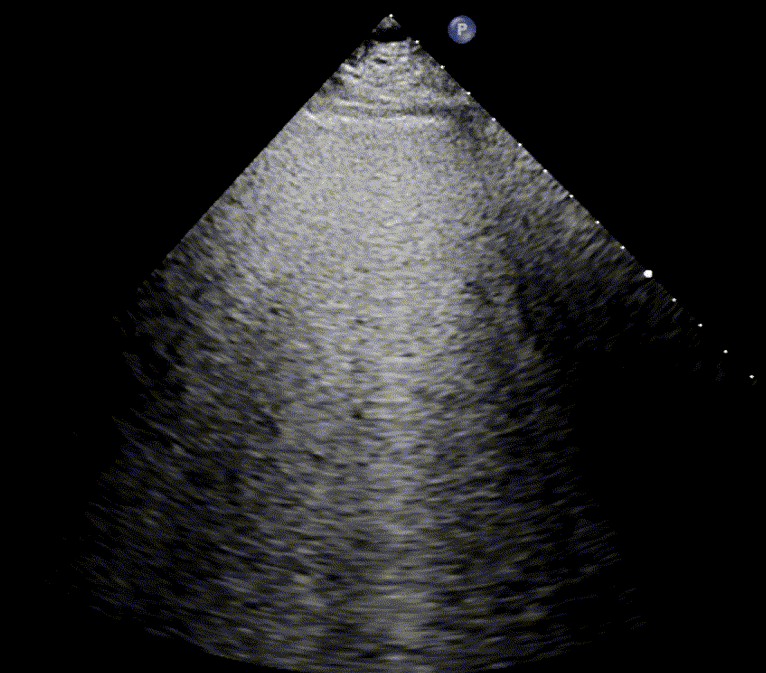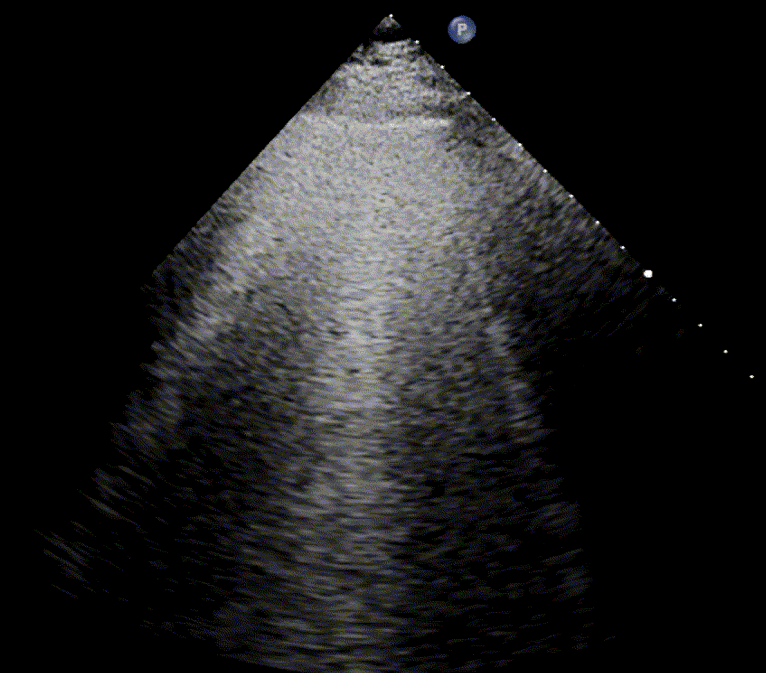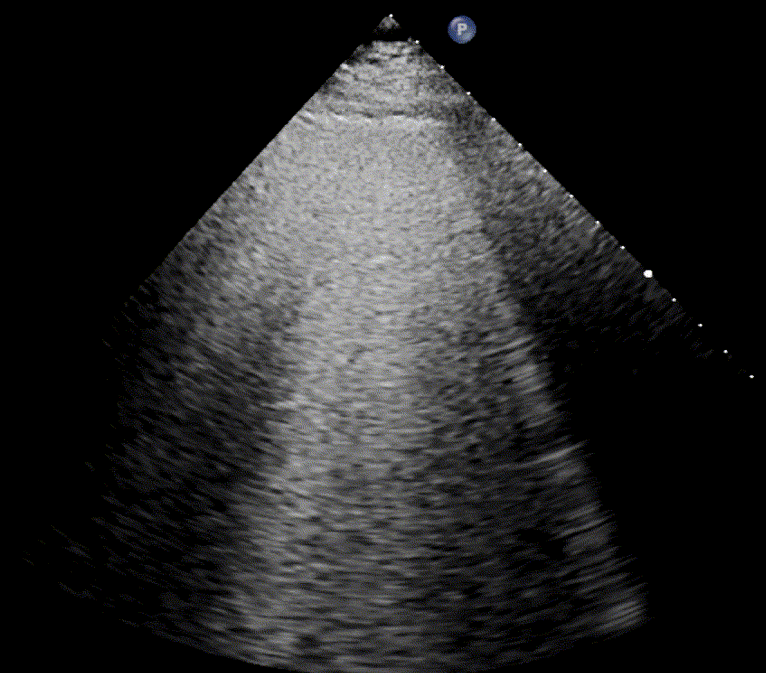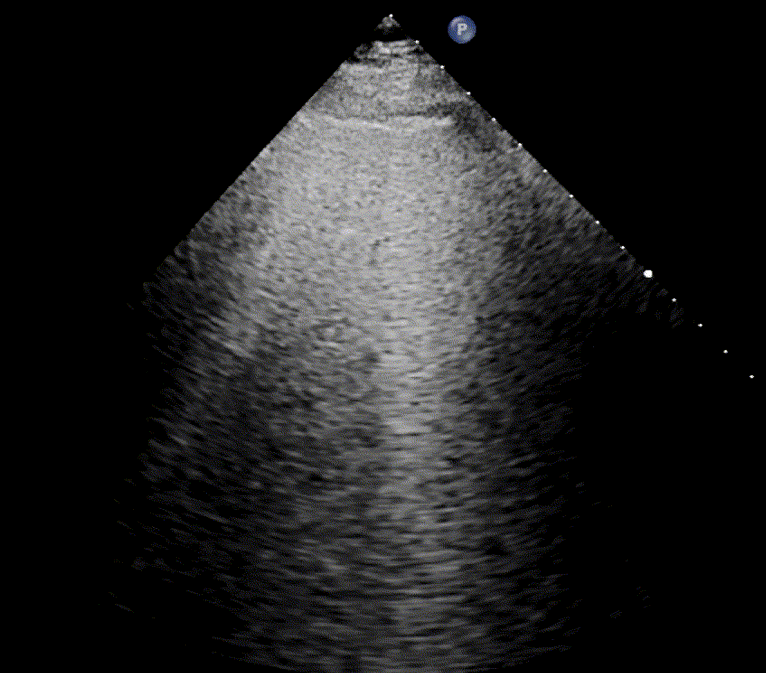
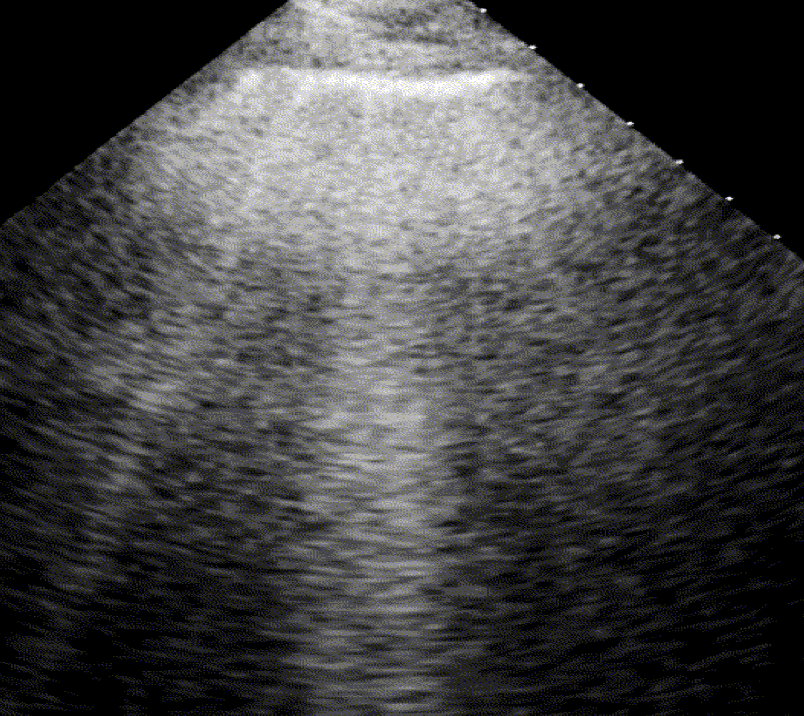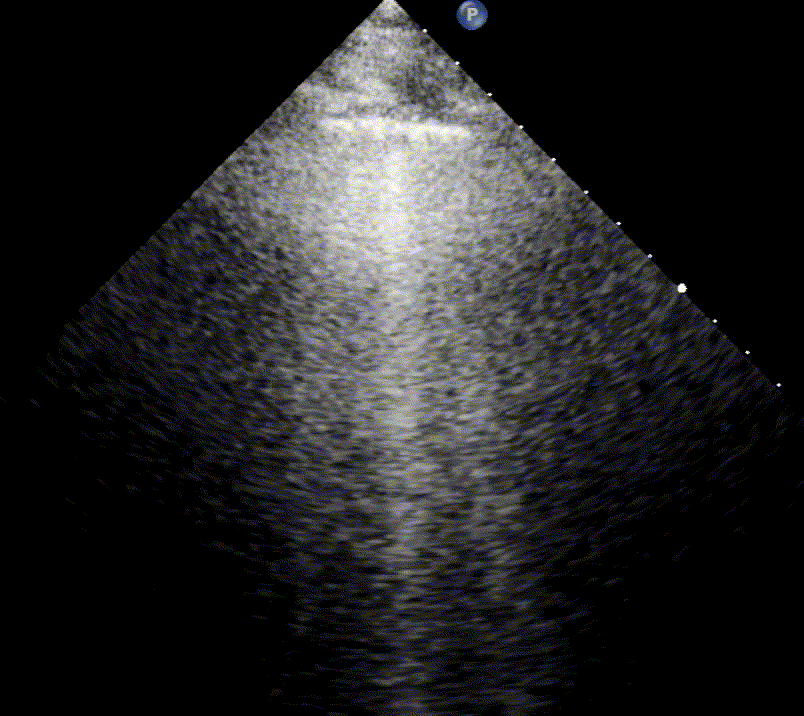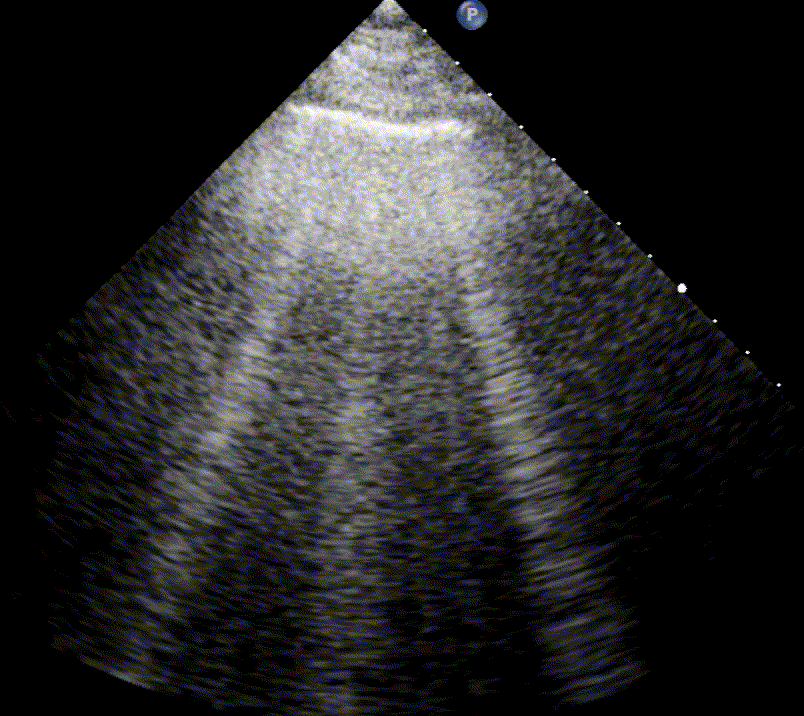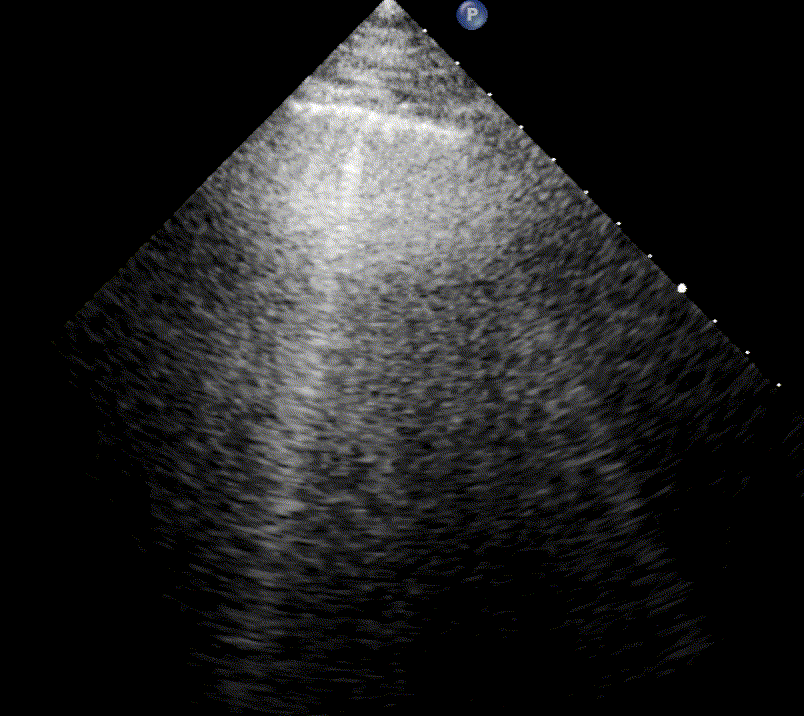
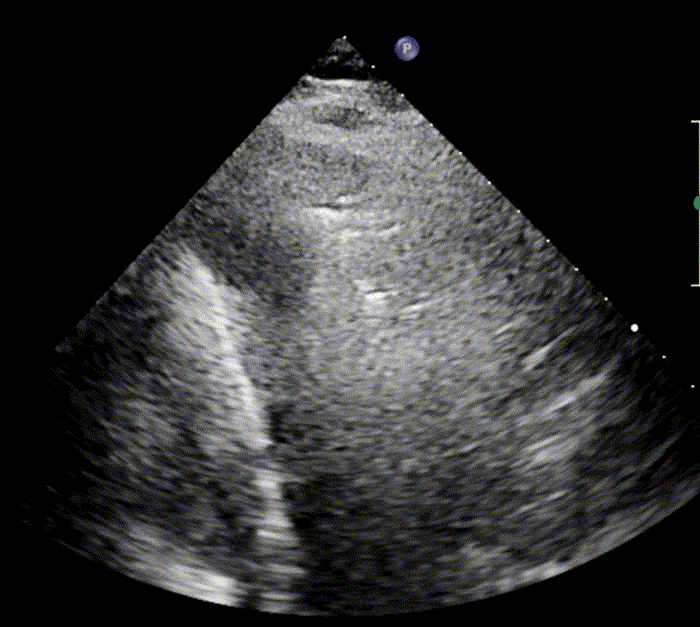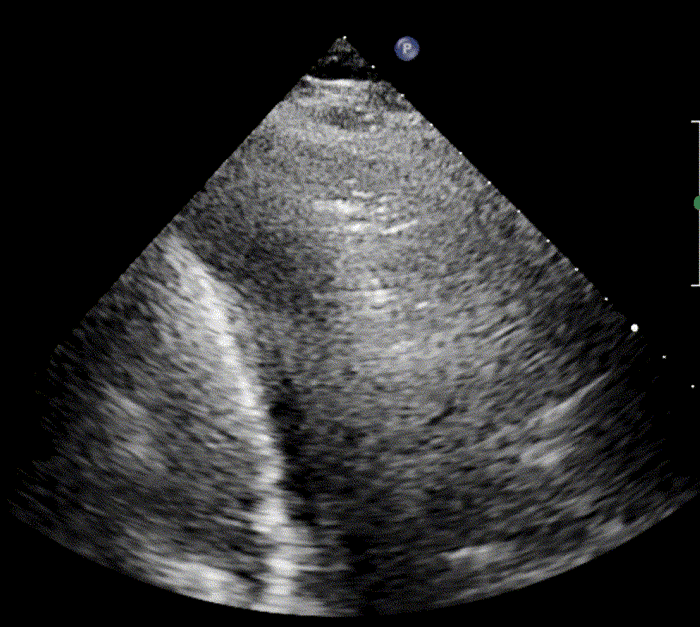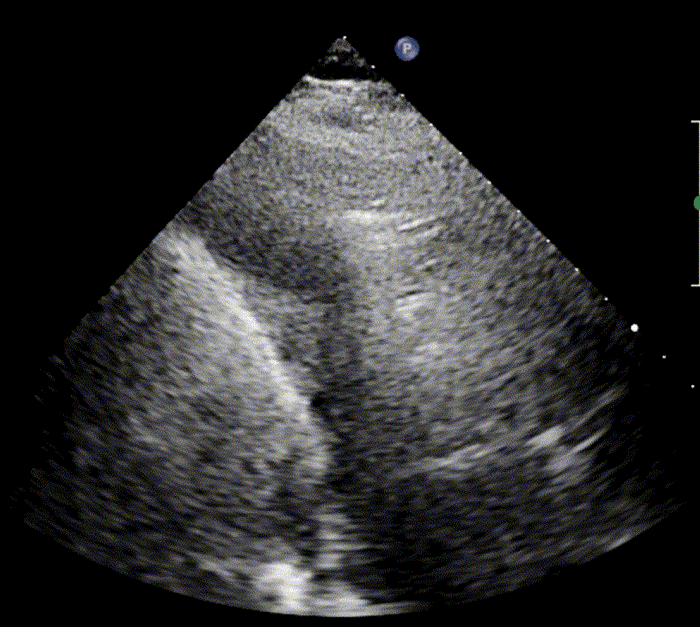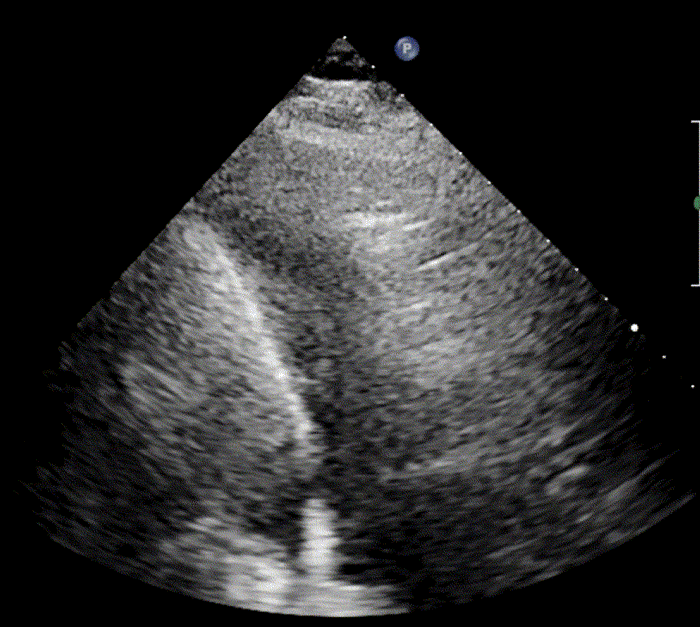
In healthy, air-filled lungs, the ultrasound image appears gray. This occurs because air scatters sound waves as they encounter solid interfaces such as the pleura, resulting in a diffuse, speckled gray appearance beneath it. Within normal lung parenchyma, there is minimal acoustic impedance mismatch to reflect the ultrasound beam; instead, the beam is absorbed by air within the alveoli. Consequently, in a normal lung ultrasound, the parenchymal texture is not visualized. The only visible structure is the pleural line — a bright, horizontal echo that moves synchronously with respiration.
As the air content of the lungs decreases, variations in acoustic properties emerge, enabling partial reflection of the ultrasound waves and the generation of visible images.
Patient Position
The patient may be positioned supine, in a lateral decubitus position, or preferably seated upright to facilitate scanning of the posterior lung regions.
In acute situations, patients are often unable to sit upright; in such cases, anterior, lateral, and posterolateral approaches typically provide sufficient diagnostic information. If a true posterior approach is required, the patient can be rolled onto their side — keeping in mind that pleural fluid will shift with changes in body position.
In the supine patient, fluid tends to accumulate first in the costophrenic angle visualized along the posterior axillary line. A more challenging method for assessing pleural effusion in a recumbent patient involves using the liver or spleen as an acoustic window and directing the probe cranially through the diaphragm to visualize the posterior costophrenic recess.
When the patient is seated, extending the arms moves the scapulae away from the posterior lung fields, further improving visualization from the back. Scanning may begin at the inferior posterior chest wall, as pleural effusions are most commonly detected in this region.
Probe Selection
Lung ultrasound can be performed using any transducer, including phased-array, linear-array, convex, and microconvex probes.
An abdominal convex probe provides a broad field of view, which often facilitates image acquisition and overall ease of examination especially for inexperienced examiners. From a cardiologic perspective, a phased-array probe is generally sufficient for comprehensive assessment.
When detailed evaluation of the pleura is required, the linear transducer offers the highest image resolution and is therefore the preferred choice for visualizing pleural structures.
Machine Setup and Image Optimization for Lung Ultrasound
When the aim is to visualize more solid tissue or fluid—such as consolidation, pleural thickening, or effusion—rather than to interpret artifacts (e.g., A- and B-lines), image optimization tools such as tissue harmonics and spatial compounding may be used to enhance edge definition and shadow clarity. In these cases, an abdominal or cardiac preset often provides suitable image characteristics.
Appropriate machine settings are crucial for accurately displaying artifacts. The ultrasound system must not filter out artifacts, as these are often the primary diagnostic features of lung ultrasound. Therefore, the default settings of modern ultrasound machines should be adjusted prior to examination to ensure optimal visualization of pulmonary findings.
For artifact imaging, select the “lung” preset if available. This preset automatically disables most image-smoothing and artifact-reduction algorithms, preserving diagnostically relevant artifacts. If a dedicated lung preset is not available, the “abdominal” preset can be used as a suitable alternative, or the “cardiac” preset if required.
General recommendations:
-Disable tissue harmonics and spatial compounding (compound imaging).
-Lower or turn off persistence.
-Set the focus (focal point) at the level of the pleural line.
-Keep the sector width and depth as small as possible to optimize the detection of lung sliding; the depth can be increased afterward if needed.
-Adjust the gain so that the pleura appears bright but not oversaturated (“burned out”).
Typical depth settings are:
-
6–10 cm when imaging the anterior lung fields
-
10–14 cm for lateral or posterior views
-
Approximately 5 cm when the goal is to visualize the pleura in detail
Optimizing Machine Settings for LUS: GE and Philips Ultrasound Systems
GE Systems
Harmonics / THI / HI / CHI: OFF. Ensure that “HI” or “CHI” does not appear in the corner of the display.
CrossXBeam / XBeam / CRI: OFF (Spatial compounding should be disabled, as it suppresses diagnostically important artifacts.)
SRI (Speckle Reduction Imaging): OFF or set to minimum
Persistence / Frame Averaging: LOW
Frequency: RES or NORMAL mode
Dynamic Range / Compression: Adjust toward moderate contrast (approximately 60–70 dB) to optimize the visibility of A- and B-lines.
Philips Systems
AutoSCAN / iSCAN: OFF
SonoCT: OFF
Harmonics: OFF
XRES: OFF
In the 2D options, check for imaging modes labeled HRES / HGEN / HPEN — these indicate that harmonic imaging is active. Turn off harmonics, you should see RES/GEN/PEN (without the “H” prefix)
How to Scan the Thorax: From Regions to the Basic Lung View
Scanning Protocols of the Thorax
The entire thorax should be examined systematically, region by region. From each region, an intercostal space that provides an optimal acoustic window should be identified for imaging. The probe is typically positioned longitudinally, oriented perpendicular to the pleural surface. When more detailed assessment of a specific intercostal space is required, the probe can be rotated into a horizontal orientation to visualize the area without rib shadowing.
The eight-zone protocol comprises four scanning regions on each side of the chest, ensuring comprehensive evaluation of both lungs.
Obtaining the Basic Lung View
Position the probe within an intercostal space and adjust its angle and placement until the “batwing sign” becomes visible. This sign appears when the acoustic shadows of the ribs and the bright pleural line together form a pattern resembling a bat’s wings. The presence of this sign confirms correct probe positioning and orientation for lung ultrasound examination.
Interpreting Lung Ultrasound: Normal and Pathological Patterns
The lungs are covered by two pleural layers: the visceral pleura, which adheres directly to the lung surface, and the parietal pleura, which lines the chest wall. These layers glide smoothly against one another during respiration. The lung interstitium forms the supportive framework of the lungs, consisting of connective tissue that surrounds and connects the airways, blood vessels, and alveoli.
Normal lung ultrasound
Ribs
Ribs are visualized as hyperechoic, curved lines that cast acoustic shadows beneath them, obscuring the underlying structures.
Pleura
On ultrasound, the pleura appears as a bright, horizontal echogenic line. For optimal visualization, reduce the imaging depth to approximately 5 cm and position the pleural line at the center of the image.
Lung Sliding / Pleural Sliding
Lung sliding (or pleural sliding) is a dynamic phenomenon observed at the pleural line, where the lung surface appears to move back and forth with respiration as the visceral and parietal pleurae glide against one another. It represents normal lung movement. This motion is often described as resembling “marching of ants”.
Any condition that separates the pleural layers or disrupts normal ventilation—such as pneumothorax, pleural effusion, or airway obstruction—will abolish this characteristic movement. In such cases, both the pleural line and the underlying lung tissue appear stationary.
Similarly, severe chronic obstructive pulmonary disease (COPD) or any process leading to lung hyperinflation may markedly reduce pleural sliding.
Importantly, absence of lung sliding does not necessarily indicate pneumothorax. Several other clinical situations can result in absent or diminished sliding, including:
-
High-frequency mechanical ventilation
-
Extensive atelectasis or consolidation
-
Pleural adhesions
-
Acute infectious or inflammatory pleural disease
-
Chemical pleurodesis
-
Advanced pulmonary fibrosis
From a clinical perspective, the presence of lung sliding reliably excludes pneumothorax at the specific site of examination, with a sensitivity of 100%.
A-lines
A-lines are highly echogenic, horizontal reverberation artifacts that appear parallel to and equidistant beneath the pleural line. They result from repetitive reflection of the ultrasound beam between the pleura and the probe, producing regularly spaced bright lines on the image.
The presence of A-lines indicates a normally aerated, air-filled lung. They are typically observed when imaging at a depth of 9–11 cm, and are most distinct when the ultrasound beam is perpendicular to the pleural surface.
A-lines may also be seen in cases of pneumothorax, as air trapped at the parietal pleura similarly reflects ultrasound waves. The key distinguishing feature is the absence of lung sliding in pneumothorax, which differentiates pathological from physiological A-lines.
B-lines
For the cardiologist, the primary application of lung ultrasound is the assessment of B-lines. These vertical, laser-like reverberation artifacts arise from the pleural line and extend to the bottom of the screen without fading. B-lines move synchronously with respiration (lung sliding), erase underlying A-lines, and share the same echogenic intensity as the pleural line.
When fluid or inflammatory exudate accumulates in the lung interstitium, ultrasound waves reverberate between air–fluid interfaces, generating B-line artifacts. The presence of more than two B-lines per intercostal space indicates that the lung tissue is no longer fully air-filled. While few isolated B-lines may be seen in healthy lungs (lung fissures may cause few B-lines), the appearance of three or more B-lines within a single intercostal space is considered pathological.
Interstitial syndrome is characterized by more than two B-lines in more than two rib spaces on both lungs. This finding reflects interstitial involvement and may occur in:
-
Acute respiratory distress syndrome (ARDS)
-
Cardiogenic pulmonary edema
-
Bilateral pneumonia or viral pneumonitis
-
Pulmonary fibrosis
B-lines are highly sensitive but not specific markers of pulmonary interstitial fluid or thickening, as they may arise from numerous pathological processes.
Pathophysiological Progression
Lung ultrasound provides direct insight into interstitial fluid accumulation. Before alveolar flooding occurs, fluid fills and thickens the interlobular septa of the interstitium. At this early stage—when alveoli are still air-filled—the sonographic pattern transitions from an A-profile (air-dominant) to a B-profile (fluid-dominant).
Notably, B-lines appear on ultrasound much earlier than Kerley B-lines on a chest radiograph and often precede clinical symptoms.
B-lines correlate with the thickening of subpleural interlobular septa in pulmonary interstitial edema.
Confluent B-lines
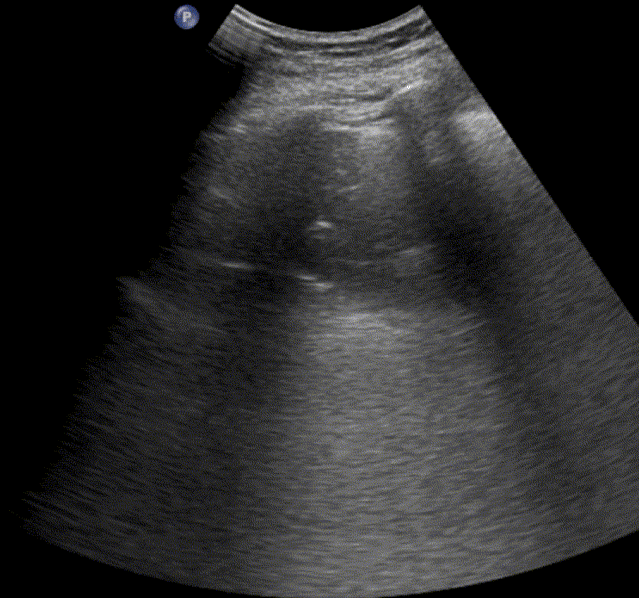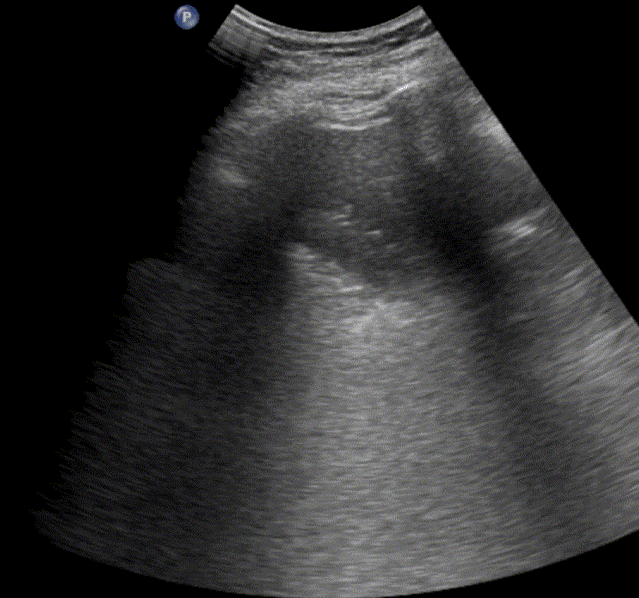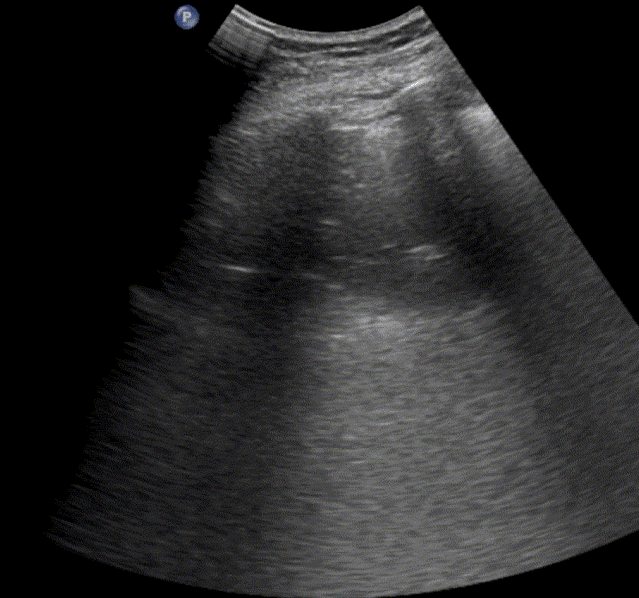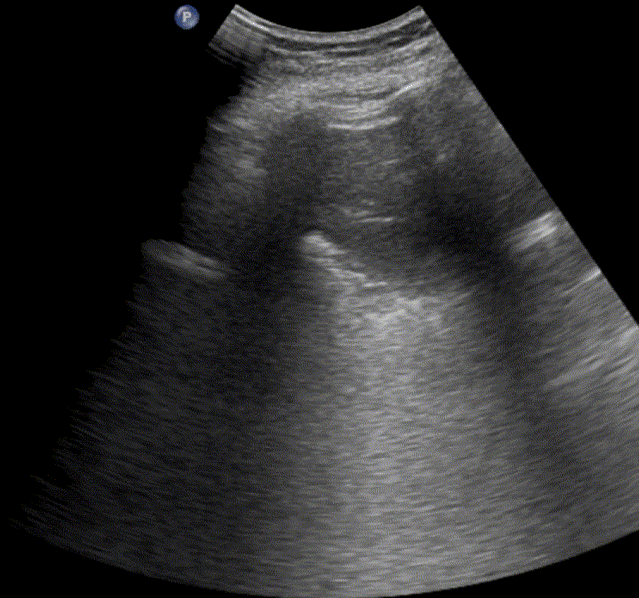
Cardiogenic Pulmonary Edema
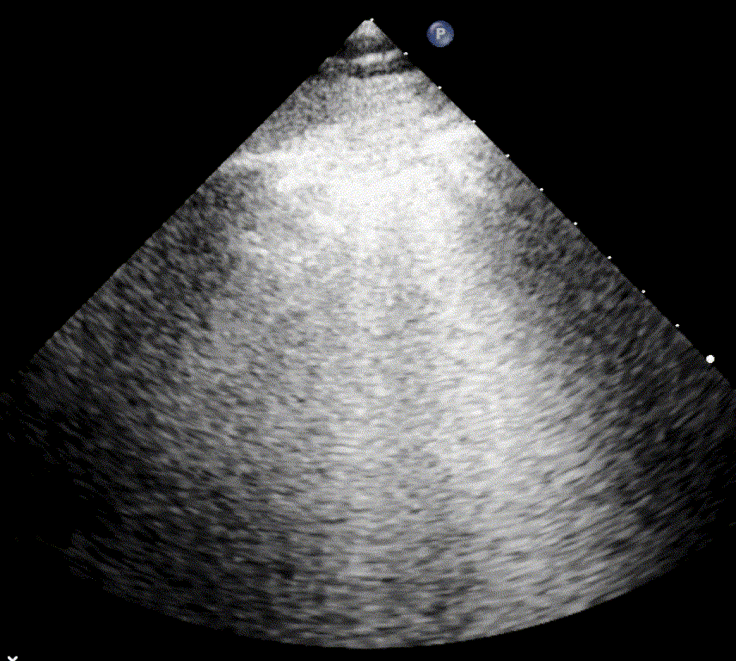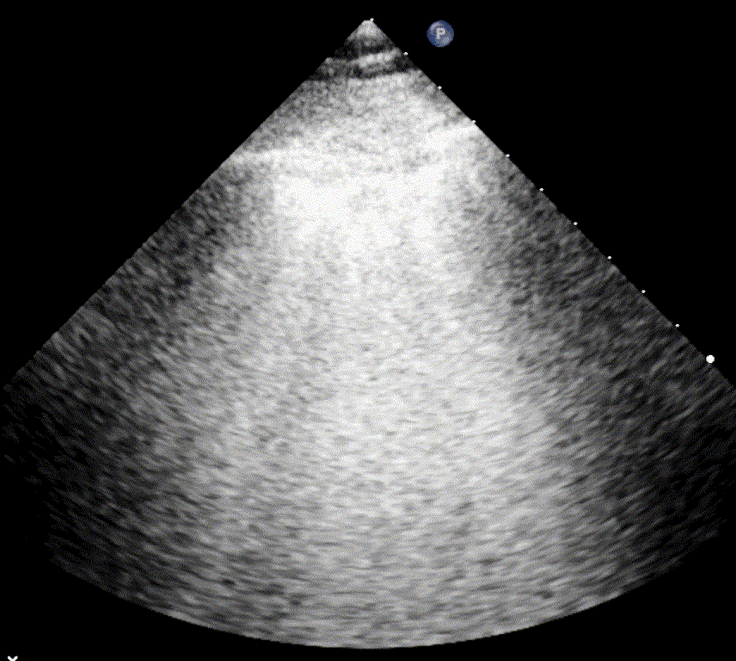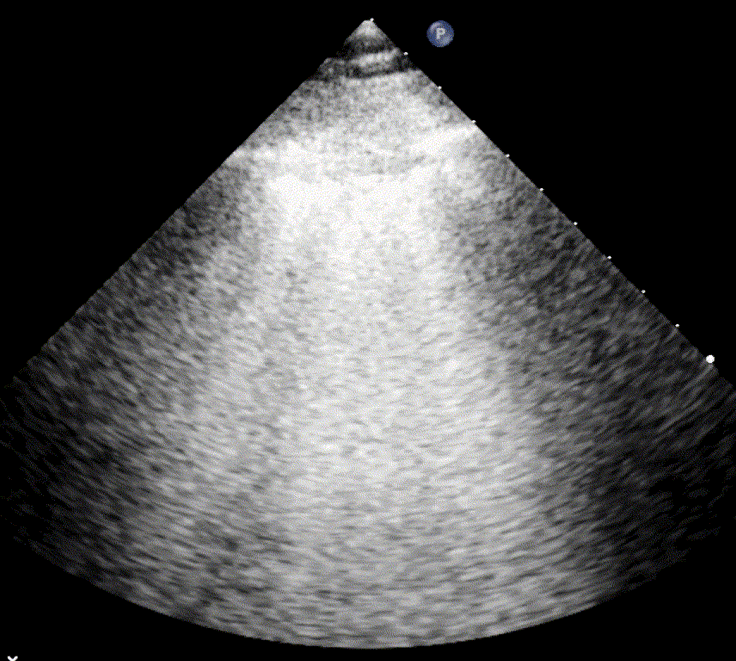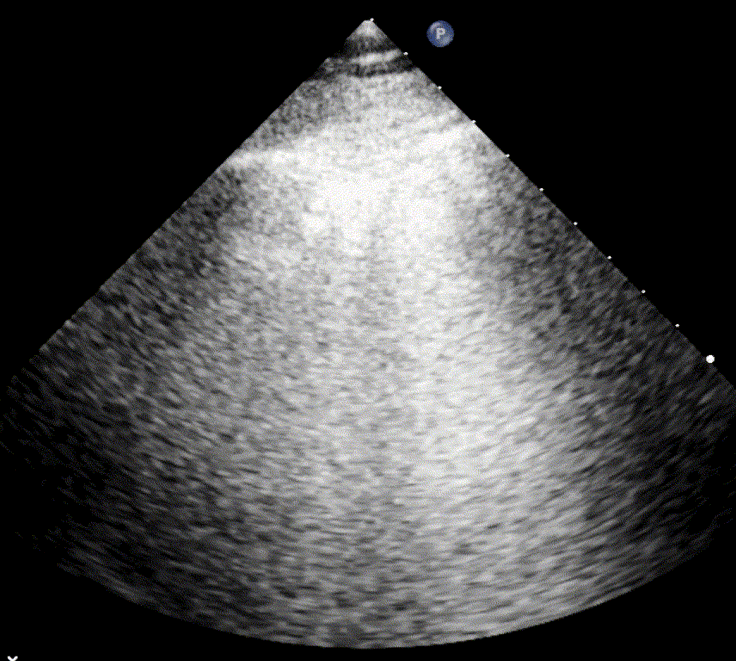
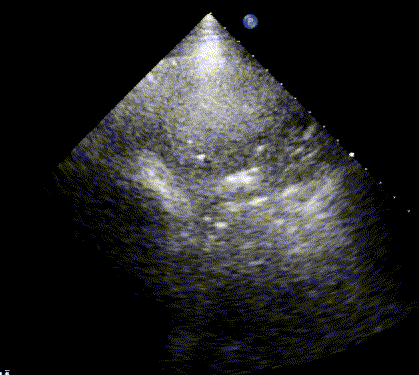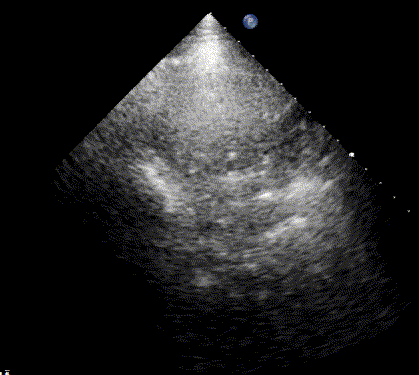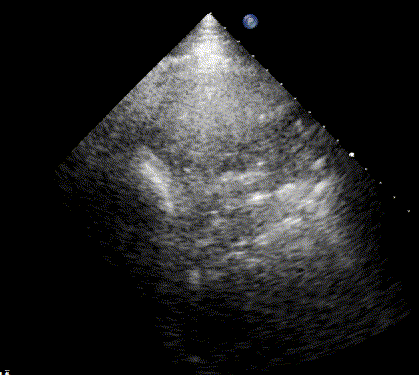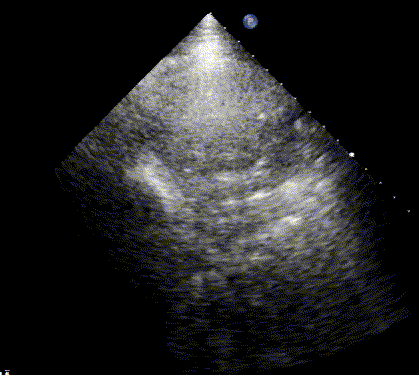
In cardiogenic pulmonary edema, the presence of three or more B-lines per intercostal space together with preserved lung sliding on both sides is characteristic. As edema worsens and air content decreases, B-lines increase in number, become broader and confluent, and may fill the entire lung field, producing an appearance of an almost uniformly bright (“white”) lung. The number and density of B-lines correlate closely with the severity of pulmonary congestion.
When air content decreases further—as in severe pneumonia or advanced alveolar flooding—the lung becomes directly visualizable as a solid, tissue-like structure resembling the liver or spleen (“hepatization”).
B-lines are very sensitive but not specific signs of cardiogenic pulmonary edema. They often appear before clinical symptoms, making lung ultrasound a valuable tool for early detection of pulmonary congestion.
Cardiogenic B-lines are bilateral, typically more prominent in the right lung, and in supine patients, tend to accumulate along the axillary lines due to gravitational effects. In such cases, pleural alterations are usually absent. The presence of multiple, diffuse, bilateral B-lines in a patient with heart failure or valvular disease strongly indicates cardiogenic pulmonary congestion.
Non-Cardiogenic and Focal B-Lines
Diffuse, bilateral B-lines may also occur in acute lung injury, ARDS, or interstitial pneumonia.
It is crucial to distinguish multiple, diffuse, bilateral B-lines—which indicate interstitial syndrome—from focal multiple B-lines, which can appear in localized lung diseases such as:
-
Lobar pneumonia
-
Pulmonary contusion
-
Pulmonary infarction
-
Pleural pathology
-
Neoplastic infiltration
In pulmonary fibrosis, B-lines arise from fibrotic thickening of the interstitium. most often fibrotic accumulation starts posteriorly at the lung bases. These B-lines are frequently associated with pleural line irregularities or thickening and correlate with the fibrotic patterns seen on CT imaging.
Patients with chronic septal scarring from prior infections or interstitial lung disease may also exhibit persistent B-lines due to permanent septal thickening or widening.
Diagnostic Implication
Although B-lines may represent various pathological conditions, their presence has one crucial implication: At any site where B-lines are visualized, pneumothorax can be confidently excluded.
Lung Ultrasound vs Chest X-Ray in Cardiogenic Pulmonary Edema
Chest radiography remains the most commonly used and widely available imaging modality for detecting pulmonary edema. However, interpretation can be challenging and subject to considerable inter-observer variability. Importantly, a normal chest X-ray does not exclude elevated pulmonary capillary wedge pressure (PCWP). According to the AHA/ACC Heart Failure Guidelines, serial chest radiographs are not recommended for evaluating pulmonary congestion in patients with chronic heart failure (HF), as their sensitivity is insufficient to detect anything other than advanced changes in lung fluid content.
In contrast, B-lines on lung ultrasound have emerged as a reliable, dynamic marker of pulmonary congestion in heart failure. Sonographic B-lines correlate well with Kerley B-lines and lung water scores on chest X-ray.
In a study by Lichtenstein et al., B-lines successfully differentiated acute cardiogenic pulmonary edema from COPD exacerbations: B-lines were present in all patients with cardiogenic edema, whereas 92.3% of COPD patients showed none, corresponding to a sensitivity of 100% and a specificity of 92%.
The diagnostic accuracy of lung ultrasound for cardiogenic pulmonary edema clearly exceeds that of chest radiography. In a meta-analysis by Al Deeb et al., point-of-care ultrasound demonstrated a sensitivity of 94% and specificity of 92% for detecting acute cardiogenic pulmonary edema. In the same review, the sensitivity of chest X-ray varied widely between 14–68%, with specificity ranging from 53–96% across three source studies.
Further supporting evidence was provided by Pivetta et al., who evaluated 518 patients with suspected acute heart failure. Patients were randomized to two diagnostic strategies:
-
Group 1: Clinical examination + bedside lung ultrasound
-
Group 2: Clinical examination + chest X-ray + natriuretic peptide (NT-proBNP) testing
In the ultrasound group, diagnostic sensitivity reached 94% and specificity 96%, compared to 85% and 89%, respectively, in the chest X-ray group.
A major advantage of B-lines is their dynamic nature. They can disappear within minutes following effective diuretic therapy, allowing real-time monitoring of treatment response. Similarly, a significant reduction in B-line number is typically observed after dialysis in patients with volume overload.
Pleural effusion
Pleural effusion refers to the accumulation of fluid between the parietal and visceral pleural layers. Because pleural fluid gravitates to the most dependent areas, it is typically detected in the lowest portions of the thoracic cavity. Diagnostic sensitivity increases when the patient is in a sitting position, as gravity allows the fluid to collect inferiorly. The lung tissue adjacent to the effusion is often compressed and partially air-depleted.
Scanning the posterolateral chest zones is most effective for identifying pleural effusions. Effusions are commonly found at the lung bases and in the costophrenic angles, where the diaphragm meets the chest wall. Compared with chest radiography, lung ultrasound is both more sensitive and more specific for detecting small volumes of pleural fluid.
Ultrasound can also be used to estimate the volume of pleural effusion. The simplest method is described by Balik et al., which involves measuring the maximal vertical separation (in millimeters) between the diaphragm and the lung base in a supine patient, and multiplying this value by 20 to approximate the pleural fluid volume: Pleural Volume (mL) = Maximal distance (mm) × 20. This formula provides a practical estimation of pleural fluid volume in clinical settings.
Loculated/Septated Pleural Effusion
Loculated pleural effusions, also referred to as septated effusions, are typically associated with exudative pleural processes. The term describes effusions in which the pleural fluid is divided into multiple compartments or “pockets” by fibrous septa.
These loculations arise from inflammatory or infectious processes, such as empyema, abscess formation, scarring, or fibrosis in the pleural cavity, which interfere with normal fluid movement and drainage within the pleural cavity.
Septated effusion in empyema
Air-bronchiogram
An air bronchogram is a pathological ultrasound finding that occurs when air remains trapped within bronchioles while the surrounding lung parenchyma becomes consolidated and loses its normal air content. The presence of an air bronchogram is highly suggestive of pneumonia or consolidation.
Dynamic Air Bronchogram
A dynamic air bronchogram is a variant of the air bronchogram sign. In this pattern, consolidated lung tissue is seen beneath the pleural line, containing bright, punctate or branching echogenic foci that move centrifugally (outward) during inspiration.
This dynamic movement of air within the bronchi during breathing is characteristic of pneumonia, indicating patent airways within consolidated parenchyma and helping to distinguish pneumonia from atelectasis, in which the air bronchogram remains static.
Tissue-like Lung Pattern
As the air content of the lung decreases, the ultrasound image begins to resemble that of solid tissue, since the absence of air artifacts allows ultrasound waves to penetrate more deeply. When all air is replaced by fluid or inflammatory material, the affected lung often appears similar in echotexture to the liver, a phenomenon known as hepatization.
This tissue-like lung pattern indicates dense consolidation and is typically associated with severe pneumonia or atelectasis. For example, the latter may result from airway obstruction (e.g., by a mucus plug) or extrinsic compression (e.g., from a large pleural effusion).
As fluid accumulation or parenchymal consolidation progresses, the ultrasound findings typically evolve in the following sequence: → Multiple B-lines → Confluent B-lines → Subpleural consolidation → Shred sign → Dense consolidation.
Pneumonia
In pneumonia, inflammation and exudation lead to the accumulation of fluid and cellular material within the alveoli and interstitium. This results in the appearance of unilateral or bilateral B-lines, depending on whether one or both lungs are affected.
Pneumonia is diagnosed sonographically by the detection of B-lines and/or parenchymal consolidation. Consolidation can be visualized by ultrasound only when it extends to or contacts the pleural surface. Very early pneumonic lesions located deep within the lung parenchyma may remain invisible to ultrasound due to intervening aerated tissue. However, in critically ill patients, consolidation reaches the pleura in approximately 98% of cases (Lichtenstein & Mezière, 2008).
Differentiating pneumonic consolidation from atelectasis by ultrasound alone can be challenging. The presence of a dynamic air bronchogram is highly suggestive of pneumonia, whereas static air bronchograms more commonly indicate atelectasis. Additionally, pleural effusion may be observed as a dark, anechoic stripe adjacent to pneumonic consolidation.
As pneumonia progresses, the number of B-lines increases, and they may become confluent. With further involvement of the lung tissue, the shred sign appears — an irregular, fragmented pleural line marking the boundary between aerated and consolidated lung parenchyma. In advanced stages, the lung may become completely airless, taking on a homogeneous, tissue-like appearance (hepatization) due to its similarity in echotexture to the liver.